CPAP ventilator design with STMicroelectronics
Follow articleHow do you feel about this article? Help us to provide better content for you.
Thank you! Your feedback has been received.
There was a problem submitting your feedback, please try again later.
What do you think of this article?
The ongoing COVID-19 crisis is sparking makers and engineers around the world into action. Everyone is considering what they can do to support the response to the pandemic.
Continuous Positive Airway Pressure (CPAP) machines are used in the treatment of COVID-19 patients where their condition doesn’t require full mechanical ventilation. CPAP devices support patient breathing by providing continuous positive pressure to the airways, ensuring that the alveoli do not fully collapse upon the patient exhaling.
CPAP devices are complex and require careful design to ensure that they can operate safely and respond to the environment in which they are operating and the unique characteristics of the patient.
Our supplier partner STMicroelectronics has worked with us to highlight how the components they provide can be used to design a safe and efficient CPAP machine. Breaking the system down into its main functional areas, we can look at the considerations that need to be made and the components we might select to meet our design requirements.
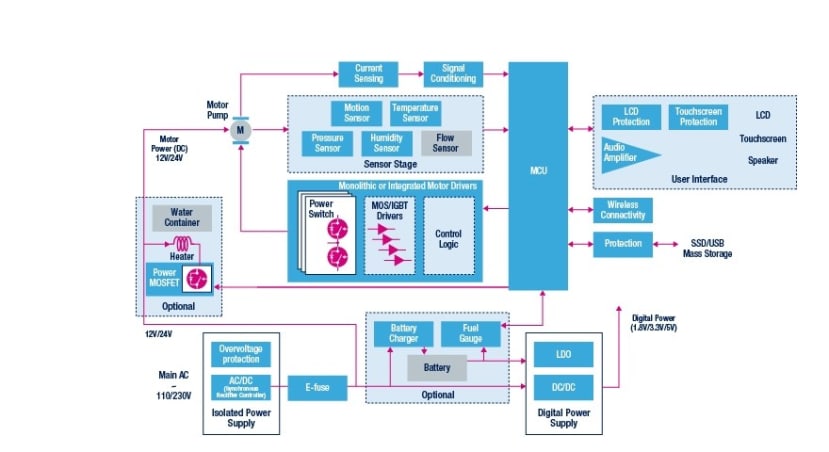
STMicroelectronics Ventilator Reference Design - Block Diagram
Sensing
CPAP systems are responsive devices, meaning they adapt their behaviour to the environment they are in and to the patient they are treating. Factors that require control include airflow rate, pressure inside the mask, temperature and humidity. In addition, external factors affecting the machine include the humidity and pressure which can vary dependent on the environment and altitude at which the machine is being used.
You may not expect altitude to have such a profound impact on the performance of a ventilation machine, however in airflow-based devices, altitude should influence motor control. At higher altitudes, the air is less dense and therefore an impeller must spin faster to deliver air pressure equal to what would be delivered at a lower altitude. For example, in Mexico City (2.2km above sea level) the atmospheric pressure is ~40kPa vs. London (11m above sea level) where it is ~100kPa.
The STMicroelectronics LPS33W barometric pressure sensor (192-4928) with integrated temperature sensor is ideally suited for this application. It can measure the airflow using the Venturi effect. This sensor is provided already packaged with gel potting and a metal enclosure to protect the sensing element from harsh environmental conditions and cleaning agents.
Temperature and humidity of the air delivered to patients must be carefully controlled to maintain optimum conditions for respiration and ensure that the airways are not dried out. The HTS221 capacitive temperature and relative humidity sensor (110-6598) provides 16-bit temperature and pressure resolution and operates across the 0-100% relative humidity range. It’s tiny 2.0 x 2.0 x 0.9mm package size makes it ideal for integration into portable healthcare devices.
Motor driver
Precise and responsive motor control is essential to maintaining the correct pressure within the CPAP mask. Different motor-control strategies can be employed to leverage sophisticated software algorithms such as the Field Oriented Control (FOC).
A great place to get started with motor control is with a motor control Nucleo pack from STMicroelectronics. ST offers evaluation kits for the L6230 series and STSPIN830 devices. The L6230 series (102-3552) of brushless motor controllers from ST is featured on the X-NUCLEO-IHM07M1 (143-8877) motor-driver expansion board. This board is also available packaged with a motor and power supply (P-NUCLEO-IHM002 (123-1061) ) to get you started. ST also offers the X-NUCLEO-IHM16M1 (182-7797) motor driver expansion board which features the STSPIN830 three-phase three-sense BLDC motor controller. This device is also available packaged with motor and power supply (P-NUCLEO-IHM03). These kits enable you to experiment with six-step and FOC algorithms, as they feature independent current sensing.
MCU and User Interface
The heart of every ventilator design is the MCU – for a basic ventilator, the STM32F302R8T6 is a great place to start and is included with the P-NUCLEO-IHM002 motor driver development kit. You can also buy the MCU development board on its own NUCLEO-F302R8 (811-2277) . You can view the full range of STM32 devices at RS Online here.
All CPAP machines require a method of communicating the current system status to medical staff and providing control over the device’s operating mode. Most modern devices use a touch-screen in combination with physical buttons to enable detailed parameters to be adjusted while allowing easy access to basic functions.
For the most complex CPAP devices, where a comprehensive UI and interfacing is required, you will need an MCU that can perform up to the task. The STM32H7 series is available with a dual-core MCU: a Cortex®-M7 @480Mhz + Cortex®-M4 @240Mhz, with an accelerated graphics capability and a crypto hash. This last feature is fundamental in medical equipment to securely maintain and transmit patient medical records.
STM32H747I-DISCO is a complete demonstration and development platform for STM32H747XIH6, designed to simplify user application development. It includes a 4” capacitive touch LCD module, it is Ethernet-compliant to IEEE802.3-2002, and features a USB OTG HS interface, SAI audio codec, ST-MEMS digital microphones and embeds the STLINK-V3E debugger/programmer.
For less complex designs you could consider the STM32F303VET6 (165-5411) 72 MHz Arm Cortex-M4 featured on the STM32303E-EVAL (864-4011) development kit. The kit is designed to showcase the full capabilities of the device, featuring two I²C, three SPI, five USART, one CAN, four 12-bit ADC, two 12-bit DAC, internal 64KB Data SRAM, 16KB Program SRAM and 512KB Flash, USB FS, and JTAG debugging and up to 24 capacitive sensing channels.
Power
Power is a key consideration for medical devices. These commonly support multiple modes of supply including mains AC input as well as rechargeable powerpack and backup battery power. This flexibility requires suitable means of switching between different power sources without disrupting the performance of the machine, battery-charge management, and AC-DC conversion that is resilient to different voltage inputs to cater to the different world markets in which the device may be sold.
You can choose to purchase a Switch Mode Power Supply off the shelf to meet your AC-DC conversion requirements. On the other hand, this may not meet your size or power requirements, so another option is to consider creating your own design to meet your specific needs. STMicroelectronics has a range of AC-DC conversion evaluation kits which offer a great starting point for your design. For example, the STEVAL-ISA147V3 (124-1179) is a 500W fully digital AC-DC power supply (D-SMPS) based on the STM32F334C8 microcontroller. The evaluation kit utilises a two-stage design: a semi-bridgeless power factor corrector (SBPFC) on the input is controlled by a STM32F051K8 (810-7500) and this feeds a regulation stage implemented as an LLC half-bridge with synchronous rectification (SR) controlled by the STM32F334C8. This design supports AC input ranging from 90V to 264V, frequencies from 45-65Hz, and provides 500W of 12V DC power.
Are you involved in medical device design and manufacture?
Let us know in the comments if you are involved in the response to the COVID-19 response in medical ventilator design.



Comments