Graphene - Unexpected Science in a Pencil Line
Follow articleHow do you feel about this article? Help us to provide better content for you.
Thank you! Your feedback has been received.
There was a problem submitting your feedback, please try again later.
What do you think of this article?
Attending the IET Yorkshire Regional Prestige Lecture 2016
Going into my second year of membership with the IET I’m always looking for cool and interesting events to attend. I had the opportunity last year as a newly joined member to attend the 2015 Regional Prestige Lecture on 3D printing, and when the 2016 Prestige Lecture came around I was keen to head along.
To begin with I wasn't quite sure what graphene was, I had an idea that it was linked to graphite, however I didn’t know how it was used or how it worked etc. I was very excited to learn more about this new material at the lecture.
Introduction
Naomi Climer, the president of the IET, gave a brief introduction before handing over to Dr. Aravind Vijayaraghavan, who is a lecturer in Nanomaterials at the University of Manchester.
Dr. Vijayaraghavan detailed what would be covered in the lecture, these points were:
- What is it?
- History
- Atomic structure
- How to make it?
- Properties
- Applications
- Beyond graphene
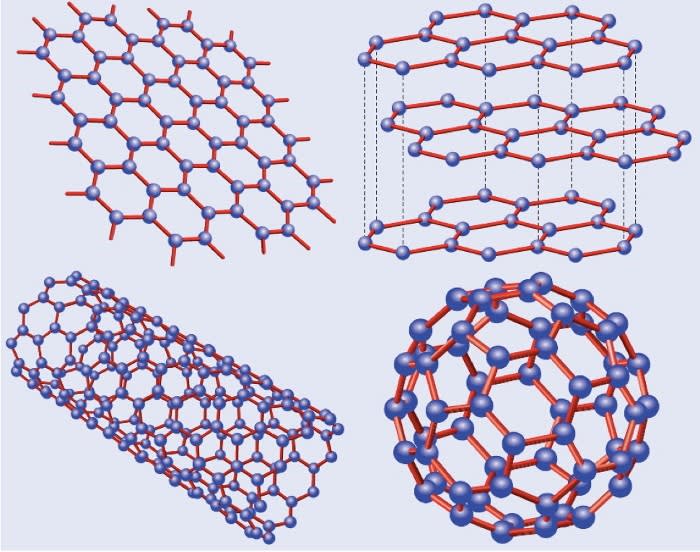
Graphene is a 2 Dimensional material which consists of a single atomic layer of graphite which is only one atom thick. The single atomic layer is purely carbon and is formed in a perfect hexagonal lattice - similar to that of honeycomb.
Source: www.slideshare.net
Carbon Dimensional Structures
Dr. Vijayaraghavan listed more common 3D structures of carbon, such as:
- Black carbon
- Diamond
- Graphite – Carbon atoms stacked in sheets
He then explained the 2D material of carbon — graphene, the 1D material of carbon — a carbon nanotube and finally (I didn't even realise this was possible) the 0D material of carbon — a Buckminsterfullerene (more commonly known as a bucky-ball or Fullerene).
Graphene can be stretched lengthways and sideways, other sheets can be added to the sides and no matter which way this happens it will always remain a single layer of atoms.
If more single layer sheets are stacked on top of this it then changes the molecular structure of the carbon atoms. This material it is no longer two dimensional as stacking these sheets make the 3 dimensional material, graphite.
A bucky-ball is made from 60 carbon atoms arranged like a football — this is made from 20 hexagons and 12 pentagons. A bucky-ball cannot be stretched or added to without becoming a completely new object all together. This means the bucky-ball has net displacement of 0, hence it is a zero dimensional object.
Source: graphene.nus.edu.sg
History and Properties
Theoretically, graphene has existed for a very long time as '2D graphite', however graphene itself wasn't discovered until 2004 by Sir Andre Geim and Sir Kostya Novoselov at The University of Manchester. 6 years later they were awarded the 2010 Nobel Prize in physics for this work, becoming the 24th and 25th Nobel laureates in the University of Manchester's history.
Most of us know that a standard pencil is made from graphite. When writing with a pencil it is layers which sheer off and leave a dark carbon trace of graphite and if this trace is thin enough you've most likely got graphene.
“If you've ever drawn with a pencil, you've probably made graphene. The worlds thinnest material is set to revolutionise almost every part of everyday life.”
Source: http://www.graphene.manchester.ac.uk
Some could say that graphene cheats nature, since it had formerly been believed that no 2D structure could be thermodynamically stable and that any movement would break the structure. Graphene is the exception to this. Due to the hexagonal lattice structure and the fact that it is not perfectly flat, it has waves and ripples on the mono-layer sheets, this means graphene can cheat nature as oscillations are simply absorbed by the structure of graphine with no physical impact on the structure whilst still remaining thermodynamically stable.
Source: www.extremetech.com
Some of the properties of graphene include:
- 200x stronger than steel and has the highest tensile strength of any material.
- Incredibly flexible and elastic
- Transparent
- Superb conductor of electricity and heat
- It can act as a perfect barrier where not even helium can pass.
- Very thin
The uses of graphene are limitless! Due to its many different unique properties this means that graphene can be used in thousands of different applications.
Production Techniques
There are several methods of extracting graphene, the first of which is the Cellotape technique, also known as Micromechanical cleavage of graphite.
The second of which is chemical vapour deposition, which involves using carbon atoms and polymers.
On a larger scale...
The third of which is one of the cheapest, however the quality of the results is poorer. This is solvent exfoliation, which forms a graphene solution.
The image of the slide from the lecture below shows each method of extracting graphene, comparing against both quality and cost.
Derivatives
Although researchers are still trying to understand the basic nature of the material, it is currently only used in a small number of potential applications. As more is learnt about the material in time it will find many more applications.
There are two derivatives of graphine, these are:
Graphane, fully hydrogenated graphene. This is remarkably stiff but stretchable and is a excellent insulator with high thermal and chemical stability.
Flourographene, a two dimensional counterpart for Teflon. This is a nanoscale replacement for Teflon, atomically thin insulator or a tunnel barrier.
New Applications
The National Graphene Institute (NGI) at Manchester is currently the worlds largest research centre and has had a huge amount of money invested into this, so that we can understand more about graphene.
Source: www.graphene.manchester.ac.uk
Indium Oxide is currently used for most touch screen technology and this will very soon run out. One of my favourite potential applications for graphene is to replace this, as it is transparent, highly conductive, lightweight and flexible this seems like the perfect alternative to me. It appears there are already prototypes being made and it would be fantastic to get my hands on one when these become available
The University of Manchester website has an amazing graphene section containing a video library explaining the material in more detail; What is graphene? How strong is Graphene? How can graphene be used in Biomedical applications? And many more!