Quantum Charging Technology: Charging at the Speed of Light
Follow articleHow do you feel about this article? Help us to provide better content for you.
Thank you! Your feedback has been received.
There was a problem submitting your feedback, please try again later.
What do you think of this article?
Adding the word ‘quantum’ before pretty much any noun or verb seems to lend a calibre of mystique unavailable from any other descriptor used in English. That may have something to do with the enigmatic behaviour of quantum level entities that make a complete mockery of the logical ‘cause and effect’ physics engineers rely on in the macro world. Alternatively, it may be simply be down to how Richard Feynman described it: “If you think you understand quantum mechanics, you don't understand quantum mechanics.”
Regardless of all that, one quantum technology that caught the imagination of tech journalists at the back end of 2022 was the concept of quantum charging. The excitement was generated by a study from scientists at the Institute for Basic Science in South Korea whose calculations suggested that quantum batteries could reduce typical home charging times of electric cars from 10 hours to just three minutes - or just 90 seconds at supercharger stations. In fact, the bigger the quantum battery, the faster it charges to capacity.
To get a handle on what’s going on with this rabidly counter-intuitive technology, we will need to go back and remind ourselves of the most basic of quantum phenomena; something you probably learned in high school chemistry.
Quantum Leap
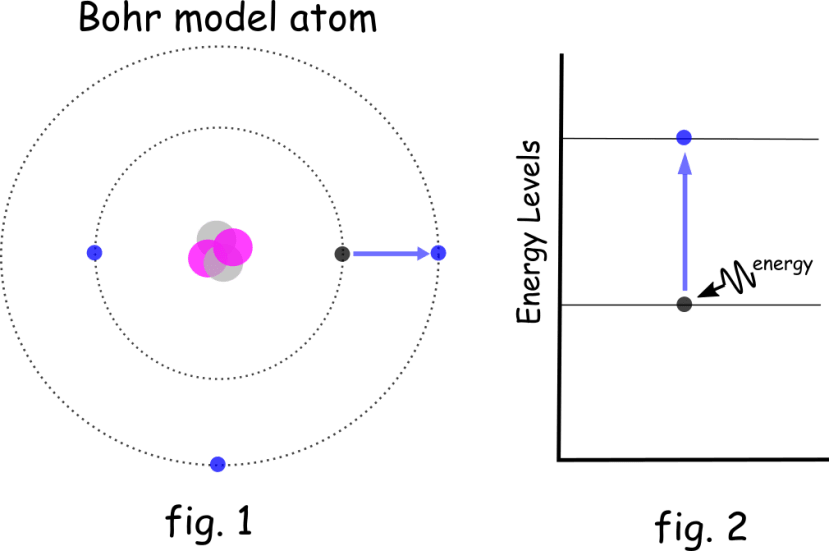
For simplicity, we will use the Bohr model of an atom (fig. 1) where we might think of electrons as analogous to planets orbiting a star, where our star is a nucleus made up of protons and neutrons:
The electron orbits (or shells) are fixed distances from the nucleus at set energy levels. Electrons at higher levels (further from the nucleus) have more energy, and their energy increases by fixed, discrete amounts. These energy levels are similar to the steps of a staircase in that you can stand on one step or the next but not in between the steps. The same for electrons: they can occupy one energy level or another but not the space between energy levels.
Electrons will always seek to occupy the lowest energy level they can but will jump from a lower level to the next higher energy level if they absorb the necessary amount of energy (fig. 2). Conversely, when electrons fall back from a higher to a lower energy level, they give off energy as photons of light. You may remember the flame test on the periodic table (group 1) alkali metals, where lithium burns red, sodium burns yellow and potassium burns lilac; this occurs when the heat energizes the electrons to jump a level and when the electron falls to its original level, the wavelength of the photon released is determined by the amount of energy between the levels. Higher energy creates higher frequency (and thus shorter wavelength) photons.
It's this increase in electron energy levels that we are using as our method for storing energy in a quantum battery.
Quantum Magic
So far, so good but how do you actually store energy in high-energy electrons when they have a habit of automatically dropping to the lowest energy level they can; and how does this lead us to ridiculously fast charging? Glad you asked, as this is where those weird quantum phenomena step to the fore.
First, we have to get energy into the electrons. One way is with photons of light. Remember how we said that an electron falling to a lower level will release a photon at a specific wavelength? Well, if we pump photons into the material at that wavelength (e.g. with a laser) then we can energise those electrons to jump up a level.
A team from the University of Adelaide, in Australia led by theoretical physicist James Quach, released the findings from their experimental study last year where they constructed a prototype battery using this exact method.
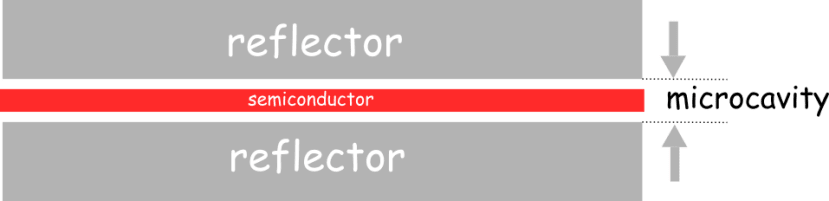
To create their proof-of-concept test device, the researchers put an active layer of light-absorbing molecules – a semiconducting organic dye called Lumogen-F Orange – into a microcavity between two mirrors made from alternating layers of dielectric materials – silicon dioxide and niobium pentoxide – to create a distributed Bragg reflector which is a mirror that reflects a lot more light than a standard metal/glass mirror.
The setup was designed to induce a quantum phenomenon known as superabsorption when laser light is fired into the microcavity. Superabsorption is a form of quantum entanglement that creates constructive interference - where two or more waves add together to produce a larger peak effect than either wave on its own - resulting in the combined molecules each absorbing light more efficiently than if each molecule were acting individually. The more energy-storing molecules there are, the more pathways exist to interfere constructively, meaning that a battery will charge faster as it is scaled up in size.
Quach's team used ultrafast transient-absorption spectroscopy with femtosecond resolution to monitor how the dye molecules were storing the energy and how fast the whole device was charging. As the microcavity size and the number of molecules increased, the charging time decreased - proving this is a viable technique for charging quantum batteries.
Researchers at Korea’s Institute for Basic Science and a group of European researchers have demonstrated similar results using micro masers: single atom masers which emit microwaves rather than visible light. As the energy output can be determined and fixed while building the micro maser, this method also ensures protection from the risks of overcharging.
So now we have our energy in the battery, how do we stabilise it so it stays in our battery until we need it? This is where it gets complicated but proposed methods include inducing dark states (where the electron ‘spin’ is magnetically modified so it no longer wants to accept energy or drop to a lower level) and a common eigenstate trapping mechanism (another technique relying on entanglement).
Conclusion
It’s safe to say that it’s a little early to be throwing out your LiON batteries. Quantum batteries were first proposed a decade ago and it has taken that long to get to proof of concept. It will take at least another decade to iron out the problems (not least of which will be determining an efficient mechanism for turning the emitted photon energy to electricity so it can power our devices) and to then commercialise the technology.
That said, this infant technology holds out some exciting prospective applications that go well beyond electric cars and consumer electronics: they could make fusion power plants, which require huge amounts of energy to be charged and discharged in an instant, a viable proposition.
This is certainly a technology to keep your eye on.